Polypropylene Structure
Polypropylene, or PP for short, is a plastic characterized by its high crystallinity and composed of various polypropylene monomers. Its exceptional toughness, rigidity, and resistance to various external factors have made it one of the most popular thermoplastics among plastic manufacturers.
As a result, polypropylene finds its way into numerous products across various industries worldwide.
In this article, I’m going to give a detailed outlook on polypropylene structure and how it affects the polymer’s general properties and other aspects
The Structure and Composition of Polypropylene
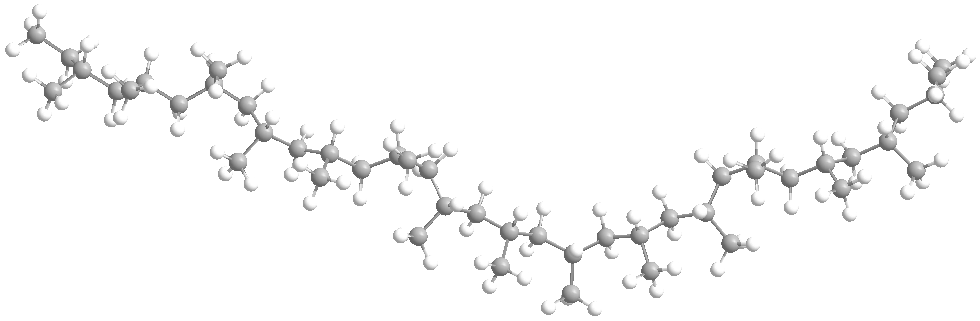
The structure of polypropylene is based on the arrangement of the repeating units along the polymer chain. Polypropylene is a linear hydrocarbon polymer consisting of repeated units of propylene monomers, and its chemical formula is (C3H6)n.
Polymerization is the process used to synthesize polypropylene, and the resulting polymer chain can vary in length, affecting the final product’s physical properties and density.
Polypropylene can be categorized into three primary types of structures: isotactic, syndiotactic, and atactic.
Isotactic polypropylene is the most prevalent, with a highly ordered crystalline structure due to the consistent direction of the methyl groups attached to the polymer backbone.
In contrast to isotactic polypropylene, syndiotactic polypropylene has a different structure. The methyl groups are arranged alternately along the polymer chain, resulting in a more flexible and amorphous structure. Due to this structure, syndiotactic polypropylene has a lower melting point and is more transparent than isotactic polypropylene. However, it is more challenging to process because of its lower crystallinity.
Atactic polypropylene is the least common form of polypropylene and has a random arrangement of methyl groups along the polymer chain. This produces a rubbery, highly amorphous polymer with a low melting point. Atactic polypropylene is not commonly known and used in commercial applications due to its lackluster mechanical properties.
How Polypropylene Structure Affects its Applications
The molecular structure of polypropylene (PP) is pivotal in determining its applicability across various industries.
The semi-crystalline nature, mainly due to the isotactic arrangement of methyl groups, imparts a balance of rigidity and toughness, making it a choice of material for applications requiring mechanical strength, like automotive parts and drones.
Its chemical resistance arises from its carbon-hydrogen backbone, making it ideal for containers holding corrosive chemicals and pipes that transport various fluids.
In the medical field, this resistance to chemical degradation is beneficial for devices that require sterilization.
PP also possesses excellent electrical insulating properties due to its low dielectric constant, making it suitable for electrical cable insulation.
These properties can be fine-tuned through copolymerization with other monomers like ethylene, which can modify PP’s flexibility, transparency, and other characteristics.
This opens up even more applications in pipes, packaging, textiles, and consumer goods like kitchenware.
Hence, the structure of PP provides the foundation for its versatility, allowing it to be tailored for an array of specialized tasks, ranging from everyday use to highly technical applications.
How Polypropylene Structure Affects its Processing
The molecular structure of polypropylene (PP) is intrinsically linked to its behavior in injection molding processes, dictating not just the material’s performance but also the settings and parameters of the molding equipment.
The molecular weight and melt flow index (MFI) of PP resins directly influence their flow characteristics.
Resins with lower molecular weight or higher MFI tend to flow more efficiently but may yield end products with compromised mechanical properties.
Conversely, higher molecular weight PP requires more force and possibly higher temperatures to fill the mold, resulting in stronger, more durable products.
The crystalline structure, primarily isotactic in commercial grades, necessitates higher barrel temperatures during injection molding, offering the benefit of increased rigidity and heat resistance in the final product.
PP copolymers, often made with ethylene, can be engineered to modify these characteristics, making the material more amenable to specific molding conditions and desired end-use properties.
The semi-crystalline nature of PP also contributes to its post-mold shrinkage and warpage tendencies, requiring careful consideration in mold design and processing conditions.
The structure of PP thus plays a critical role in determining its suitability for specific injection molding applications and requirements, impacting everything from machine settings to cycle times and, ultimately, the quality of the molded product.
Molecular Weight and Blending of Polypropylene
Polypropylene’s molecular weight is the total weight of all the monomer units in the polymer chain. Higher molecular weight polypropylene has greater mechanical strength and stiffness but is more challenging to process.
Lower molecular weight polypropylene is more flexible and easier to process, but its mechanical properties are inferior.
PP can also be blended with other polymers to alter its thermal properties.
By blending PP with HDPE, for instance, its flexibility and impact resistance can be improved.
Summary
To sum up, polypropylene’s structure significantly impacts its properties, and its classification based on molecular weight and blending provides valuable insights into its behavior in different applications. Additionally, the capability to blend polypropylene with other polymers presents promising opportunities for developing innovative and enhanced products.
Quick Navigation